WhiMSICAL Registry Frequently Asked Questions (FAQs)
FAQ Introductory

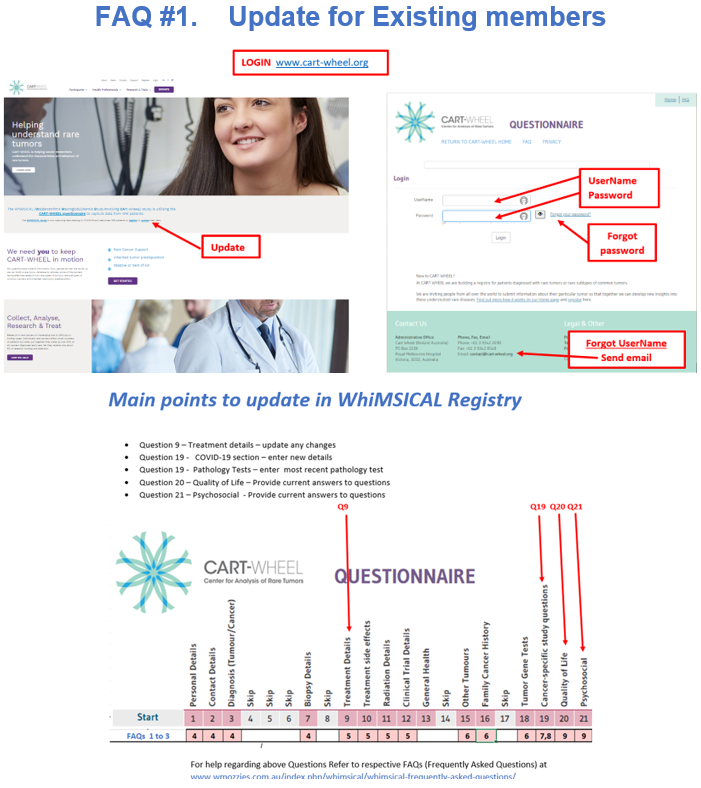
FAQ #2 Registration and Consent




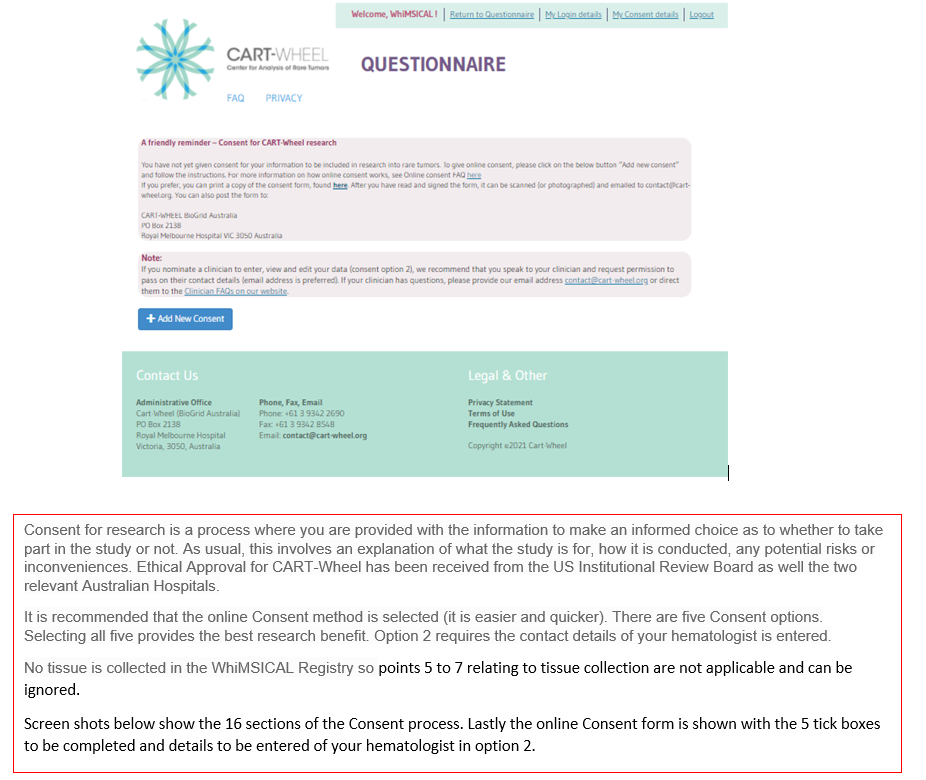

Points marked about Tissue Collection may be ignored as there is no tissue collection involved in CART-Wheel.




For this study, there are five different levels of consent. It is recommended that all five be selected and that in option 2 details of your hematologist be provided.

FAQ #3 General tips for completing questionnaire:
- How to save, logout and return later
- Making use of the “Tip” boxes and hovering over the underlined words
- Navigating through the questions using the banner
- Questions to skip once you have entered WM as diagnosis.

FAQ #4 Questions 1 to 3, and 7
- Filling out personal details. This is done to ensure the study is conducted correctly, with each person’s details linked to their consent. It is also to ensure there are not duplicate entries for data analysis. All information is stored very securely, kept confidential and is not shared with any third party outside of an ethically-approved research project. WhiMSICAL study investigators can only access your de-identified data to increase the layers of confidentiality. For an additional layer of security, personal details are stored on a separate server to the clinical data entered in the rest of the questionnaire.

- Q3: Enter “Wa” and select Waldenstrom’s Macroglobulinemia from the drop-down menu.
- For completing the bone marrow biopsy results, don’t worry if your report does not have the specific information asked here. Fill in what you have.
- If you are unsure of the specific date of diagnosis, hover over “Estimated date” for an explanation of how to complete it

Once you click on save and next, a welcome to the WhiMSICAL project will pop up and the link will take you to the next relevant question -> Q7
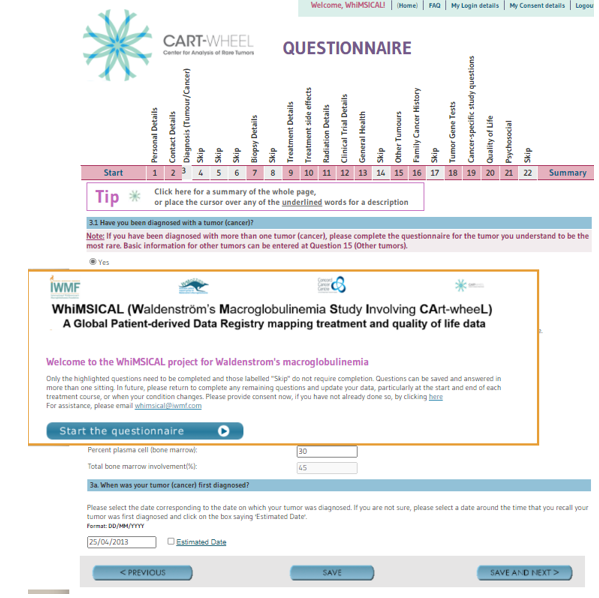

FAQ #5 Question 9-12, completing treatment details:
- Needs just going through with share screen
- Additional points for discussion:
- Enter clinical trial at Q9 and in Q12
- Treatment data is the most important to help researchers determine the effectiveness of different therapies and treatment practices
- If you are unsure of the specific date of treatment, hover over “Estimated date” or an explanation of how to complete it
- Additional points for discussion:


FAQ #6 Questions 15,16 and 18, additional cancers, family history and tumor gene test
- These questions are to about risk factors for developing cancer and to learn more about how different types of WM present and respond to different treatments.

Q18: this is specifically about special gene tests that were performed on your tumour biopsy or bone marrow biopsy, not a test to see if you carry a gene that puts you and your family at risk of cancer. Typical tests for WM include MYD88, CXCR4, “cytogenetics”, next generation sequencing.

FAQ #7 Q19 COVID questions

FAQ #8 Q19 Symptoms and pathology:
- Key timepoints are diagnosis, before 1st cycle and after last cycle of treatment. If on continuous therapy, e.g. ibrutinib, then complete every 3-6 months. If you have never had treatment, yearly results or whenever your symptoms change.






FAQ #9 Q20 and 21: QoL and psychosocial questions:
Key timepoints are diagnosis, before 1st cycle and after last cycle of treatment. If on continuous therapy, e.g. ibrutinib, then complete every 3-6 months. If you have never had treatment, yearly results or whenever your symptoms change.







